More Information
Submitted: January 30, 2024 | Approved: February 27, 2024 | Published: February 29, 2024
How to cite this article: Saiz-Figuerez MI, Nava E, Galindo MF, Jordan J. Is there a Place for Klotho in Alzheimer’s disease? J Sports Med Ther. 2024; 9: 001-010.
DOI: 10.29328/journal.jsmt.1001074
Copyright License: © 2024 Saiz-Figuerez MI, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Alzheimer´s disease; Klotho; Neurodegenerative aging; Aβ clearance; FGF23; TGF-β; NF-kB; IGF-1; Wnt; Anti-aging
Is there a Place for Klotho in Alzheimer’s disease?
Maria Isabel Saiz-Figuerez1, Nava E2, Galindo MF3 and Joaquin Jordan1*
1Área de Farmacología, Departamento de Ciencias Médicas, Facultad de Medicina de Albacete, Universidad de Castilla-La Mancha, Albacete, España
2Área de Fisiología, Departamento de Ciencias Médicas, Facultad de Medicina de Albacete, Universidad de Castilla-La Mancha, Albacete, España
3Área de Tecnología Farmacéutica, Departamento de Ciencias Médicas, Facultad de Farmacia, Universidad de Castilla-La Mancha, Albacete, España
*Address for Correspondence: Joaquin Jordan, Área de Farmacología, Departamento de Ciencias Médicas, Facultad de Medicina de Albacete, Universidad de Castilla-La Mancha, Albacete, España, Email: joaquin.jordan@uclm.es
Alzheimer’s disease, a major healthcare concern, lacks an effective pharmacological therapy to change its irreversible progression. In this work, we present Klotho, a protein associated with aging that is involved in the regulation of numerous physiological processes and is a serious candidate to be a pharmacological target to act on. Klotho’s mRNA has been found in neurons of a variety of brain regions (cortex, hippocampus). The best studied and prominent function of Klotho is as the co-receptor of fibroblast growth factor 23 (FGF23), through which Klotho controls renal phosphate excretion and vitamin D metabolism. Reduced serum levels of Klotho in mice have been associated with a shorter life expectancy and with numerous pathological conditions such as renal disease, vascular calcification, neurodegeneration, and others. Moreover, overexpression of Klotho leads to opposite effects resulting in increased survival rates. In this review we address different signaling pathways in which Klotho is involved in one way or another, focusing on those pathways that could serve as pharmacological targets to modify the evolution of Alzheimer’s disease. We describe how Klotho inhibits signaling cascades involved in cellular senescence, fibrosis, inflammation, and apoptosis all of which are mediated by tumor growth factor β (TGF- β), nuclear factor kappa K (NF- κ B), insulin-like growth factor 1 (IGF-1) or Wnt. We also highlight how Klotho is able to activate anti-inflammatory and antioxidant signaling pathways. Although there are no drugs that act specifically on Klotho, compounds currently on the market such as hormone-based drugs, pravastatin, losartan, fosinopril, and rapamycin have been shown to increase the expression of this protein and are also discussed.
When Aloise Alzheimer first described, back in the early 20th century, the anatomopathological abnormalities of Auguste Deter’s brain, he was unaware that he was opening the doors to the field of clinical research on dementias: a family of diseases, in addition to Alzheimer’s Disease (AD) itself, that includes others such as vascular dementia, Lewy body dementia, and frontotemporal dementia. The total number of new cases of dementia worldwide is increasing year by year. For example, between 1990 and 2019, AD prevalence rose from 2.9 million to 7.2 million, an increase of 147.7%, and is expected to affect more than 150 million people by 2050 [1].
Unfortunately, we currently have no effective pharmacological therapy capable of reversing the undesirable and irreversible evolution of AD. Possibly, the main cause of this failure is that, in many cases, AD is diagnosed in late stages, when the potential for improvement is very small. AD can be broadly classified into two major classes: familial and sporadic AD. The familial form is associated with mutations in three genes: Amyloid Precursor Protein (APP), presenilin 1, and presenilin 2. On the other hand, the sporadic late-onset form has been related to environmental factors such as the presence of heavy metals (e.g. lead, manganese, and cadmium), and other metals like aluminum and smoking. In addition to this, there are numerous genetic factors such as the Apolipoprotein E (APOE) gene and its gene variants Epigenetic mechanisms are also involved. Nevertheless, it is clear that the most important factor associated with sporadic AD is aging.
Aging, like all multifactorial physiological processes, is genetically regulated [2] and is characterized by disorders, often irreversible, of different tissues and organs [3] causing physiological dysfunctions that can lead to a state of disease. Different transcription factors and proteins have been associated with the programming machinery of aging such as p53, telomerase, and Klotho.
Klotho is a beta-glucosidase implicated in the control of autacoid-mediated functions. The history of klotho is linked to a paper published in 1997 where Makoto Kuro-o described a transgenic mouse with age-related disorders caused by a transgene insertion mutation. Homozygous mice showed phenotypes similar to those of patients with premature aging syndromes: arteriosclerosis, osteoporosis, age-related skin changes, and ectopic calcifications, as well as short lifespan and infertility. The authors named this mutant Klotho in honor of one of the Fates, the Greek goddess who weaves the thread of life [4].
The Klotho family, named Kl, has three members, including α-Kl, β-Kl and γ-Kl. In general, the word “Klotho” refers to α-Kl when no subfamily is mentioned [3,5]. The human Kl gene (α-Klotho) is located in chromosome q13.1 and consists of five exons flanked by PDS5B and STARD13 [6]. The promoter region is rich in Sp1 and cooperates with Oct-1 [7] enhancing gene expression. There are five exons covering 50 kb on chromosome 13q12 [8], and four introns in the KL coding region transcribing the mRNA of 3036, 3042, and 3042 nucleotides respectively. The Klotho gene has a functional variant, known as KL-VS (V allele), which contains three coding variants, two amino acid substitutions (F352V, C370S), and one silent mutation (K385K). Homozygosity for the V allele is associated, in humans, with reduced longevity and both heterozygotes and homozygotes have an increased risk of early-onset coronary artery disease [1,2,8].
The α-Klotho has two alternatively spliced variants, a membrane form of 1012 amino acids and a secreted form of 549 amino acids. The latter, more abundant, lacks the second internal repeat, the transmembrane domain and the intracellular domain of the membrane form. It has 1012 amino acids with a very short intracellular C-terminal sequence (10 amino acids), a transmembrane domain, and, for the most part, an extracellular portion [1]. Extracellularly, comprises two soluble domains: in its proximal part, KL1, and in its terminal part, KL2, which can be cleaved by membrane proteases (ADAM10 and ADAM17). Klotho can be released to blood, urine, and cerebrospinal fluid as a soluble form (named α-Klotho or s-Klotho). When released into the blood, s-Klotho acts as an endocrine hormone. Subsequently, s-Klotho can be generated directly through alternative RNA splicing or proteolytic cleavage [5].
There are also soluble forms of Kl (s-Kl) that can be produced not only by shedding the extracellular domain of Kl through the proteolytic activities of disintegrin and metalloproteinases 10 and 17 (ADAM10/17). However, it can also be formed by alternative splicing of the Kl gene.
The main source of Klotho is the kidney [9,10] but it is also expressed in the parathyroid glands as well as in the pancreatic β cells, blood vessels, ovary, testis, inner ear, skin peripheral blood circulating cells and central nervous system [11,12]. As stated above, the soluble forms are mainly found in body secretions such as blood, urine, and Cerebrospinal Fluid (CSF) and have endocrine, paracrine, or autocrine roles that are independent of growth factors [13].
Klotho-regulated pathways
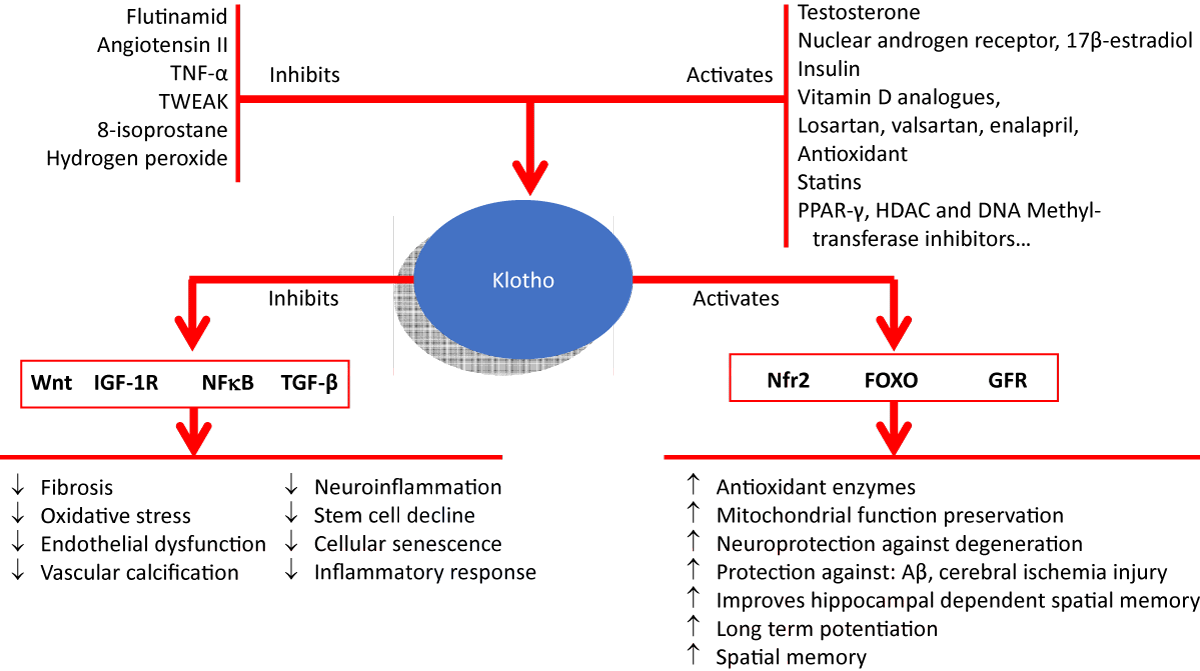
KLOTHO protein is a beta-glucosidase involved in the regulation of a wide variety of physiological functions modulated by autacoids such as fibroblast growth factor-23 (FGF23; phosphate, calcium and vitamin D excretion), transforming growth factor beta (TGF-β; senescence, fibrosis), insulin growth factor (IFG-1), Wnt/-catenin (tissue fibrosis), nuclear factor (erythroid-derived 2)-like 2 (Nrf2; antioxidation and autophagy) and nuclear factor kappa B (NF-κB; free radicals and oxidative stress) (Figure 1).
Figure 1: Comprehensive figure depicting the known hormone-, antioxidant- and antiinflammatory-based pharmacological pathways that act on the regulation of Klotho levels and the associated downstream signaling pathways with the final effects.
The extracellular domain of the transmembrane form of Klotho acts as a co-receptor of the FGF-23 receptor (FGFR). By working as, a co-receptor the affinity binding of FGF23 to FGFR1c, FGFR 3c as well as FGFR 4c can increase up to 20-fold [11,13-16]. FGF23 is synthesized mainly in the bone tissue [11] and participates in the regulation of phosphate and vitamin D balance [2,3,5,14,17]. Upon activation of FGFR, a signaling pathway is initiated through PI3K/Akt, phospholipase C γ (PLCγ), and Ras/MAPK/ERK [13,18,19].
Another pathway related to Klotho is the TGF-β signaling pathway which appears to be inhibited by this protein. TGF-β is a pleiotropic cytokine that binds to a receptor comprised of type 2 TGF-β receptor (TR2), and TβR1. TGF-β is involved in the development of cellular senescence, stem cell decline, immune impairment, and other alterations associated with aging [20-23].
IGF-I pathways have also been linked to Klotho. s-Klotho, by inhibiting IGF-I signaling and that of its receptor (IGF-IR) [24], contributes to the amelioration of aging [25]. IGF-1 is a growth factor with effects on development, growth, cell differentiation, and tissue repair [26]. Insulin/IGF-1 receptors are transmembrane tyrosine kinases that, upon ligand binding, initiate the signaling process by phosphorylation and protein binding of the insulin receptor substrate (IRS) [27]. Activation of s-Klotho connects antioxidant mechanisms through FoxO forkhead (FOXO) transcription factors. Blockade of insulin/IGF-1 pathways unlocks inhibition of these transcription factors, leading to their nuclear migration and to the expression of antioxidant enzymes, such as manganese superoxide dismutase [28].
Klotho blocks Wnt, a family of lipoproteins secreted upon Klotho’s binding to several ligands which include Wnt1, Wnt3, Wnt4, and Wnt5a [29-31]. Klotho regulates antioxidant pathways via the nuclear factor (erythroid-derived 2)-like 2 (Nfr2). This factor regulates the expression of genes involved in the protection against oxidative stress and inflammation, in addition to vital actions such as mitochondrial function preservation, protein homeostasis, autophagy regulation, and damaged protein elimination [32-34].
Additionally, Klotho blocks the signaling pathway of NFκB. This transcription factor is involved in the immune and inflammatory response as well as other cell-critical processes, such as anti-apoptotic signaling, proliferation, and oxidative stress [35-37]. Klotho carries out this modulation directly, by preventing the translocation of the NF-κB subunit Rel A (p65) from the cytoplasm to the nucleus, or by preventing the degradation of the IκB protein [38]. Thus, it has been shown that α-Klotho is able to decrease NF-κB activation and to reduce the expression of IL-8, MCP-1, RANTES, and IL-6 as well as the expression of adhesion molecules such as intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1) [39,40]. Moreover, the existing negative feedback between NFκB activation and the expression of Klotho should not be overlooked. Indeed, activated NFκB is capable of inhibiting Klotho, and under conditions of high inflammation, the expression and activation of NFB is higher than that of Klotho leading to the suppression of the latter [41,42].
Klotho and pathophysiology
Klotho and KL play important roles in aging-related disorders, such as chronic kidney disease [43], cardiovascular diseases [44], diabetes [45], cancer [46], and neurological disorders. A higher circulating level of Klotho is associated with a lower risk of metabolic syndrome, renal disease, and cardiovascular disease [5], and its overexpression results in increased survival [1]. On the other hand, the serum level of this protein decreases with age and is inversely related to aging phenotypes and other conditions such as renal disease, vascular calcification, cardiac hypertrophy, hypertension, fibrosis, osteopenia, pulmonary disease, neurodegeneration and, ultimately, to a higher mortality rate and shortened longevity [1]. In animal models of disease, a lack of Klotho in the brain has been associated with cognitive impairment, premature death, and synaptic loss [47]. Specifically, the human KLOTHO protein has 86% amino acids in common with murine KLOTHO. Mice deficient in Klotho develop cognitive deficits such as memory impairment and hippocampal damage.
Pharmacological regulation of Klotho levels
Several transcription factors are involved in the regulation of Klotho expression: activators, such as PAX4 [48], Sp1 and Oct-1, vitamin D, PPAR-γ [49,50], and inhibitors like FGF23 [51,52], epidermal growth factor, erythropoietin, the ras homologous gene of family A, AP-2, E-box, and NF-κB [1,53]. By operating on these regulatory factors it is feasible to pharmacologically modulate Klotho and thus achieve therapeutic benefits from this protein. We will briefly describe some of the drugs capable of regulating the Kl gene:
Hormone-based drug therapy: This has a huge potential and can be considered a modulator of Kl expression and therefore should be regarded as a first-level strategy to take advantage of Klotho’s properties. Kl expression can be modulated by numerous hormones (both at the level of the membrane and the secreted forms). For example, testosterone [54], upregulates mRNA and Kl protein levels in NRK-52E cells. Positive regulation of nuclear androgen receptor (AR) upregulates Kl levels. Flutamide, an AR antagonist drug, attenuates testosterone-modulated Kl expression. 17β-estradiol activates Kl in the hippocampus [55], while triiodothyronine increases the expression of the membrane, but not the secreted, form of Kl in 3T3-L1 adipocytes [56]. Finally, insulin enhances s-Kl production through a phosphoinositide 3-kinase (PI3K)-dependent pathway in which the ADAM 10/17 protease is involved [57].
The binding of the pleiotropic steroid hormone vitamin D to its superfamily of nuclear hormone receptor transcriptional regulators is involved in the positive regulation of Kl [58-60]. Vitamin D analogues including calcitriol, alfacalcidol, doxercalciferol, fluorocalcidol, and maxacalcitol, could have inducing effects on Kl gene expression. In this sense, calcitriol or its analog, paricalcitol, has been shown to elevate serum Kl levels in mice, independently of parathyroid hormone and calcium level alterations [58].
Inhibitors of the renin-angiotensin system: Long-term administration of Ang-II reduces the renal level expression of Kl mRNA and proteins [61] by a mechanism involving transforming growth factor-β1 (TGF-β1)- p38 MAPK-P53-SP1 and resulting in the binding of P53/SP1 to the Kl gene promoter inhibiting its transcription [61]. Thus, angiotensin receptor antagonist drugs, such as losartan and valsartan, and ACE inhibitors, such as enalapril and fosinopril, could increase Kl levels [62].
Anti-inflammatory agents: Anti-inflammatory drugs, steroidal and non-steroidal, can be effective in maintaining or even elevating Kl expression. Some inflammatory cytokines such as tumor necrosis factor α (TNFα) or the weak inducer of TNF-like apoptosis (TWEAK) reduce Kl expression [41]. The effect of TNFα appears to be mediated by a nuclear factor kappa (NF-κB) -dependent mechanism. Whereas TWEAK inhibits Kl gene expression, by inducing RelA binding to the Kl promoter, causing its deacetylation [41].
Antioxidants: In conditions where oxidative stress is evident (elevations of 8-isoprostane or hydrogen peroxide), Kl expression is inhibited [63]. In these cases, supplementation with antioxidants could be considered as another therapeutic approach. Vitamin C, vitamin E, N-acetylcysteine, melatonin, lipoic acid, and polyphenols such as curcumin, could lead to an increase in Kl [64,65]. Interestingly, the antioxidant properties of Klotho have been unintentionally used for ages. Indeed, natural alkaloids, organic compounds that contain nitrogen and constitute one of the most important effective ingredients in Chinese traditional herbal medicine, upregulate the expression of Klotho as shown by Rui, et al. [66]. These authors propose that the protective role of the herbs would be mediated by antioxidant mechanisms, mitochondrial damage improvement, cell death reduction, and inflammation inhibition. For instance, one of these alkaloids, neferine, a bisbenzylisoquinoline alkaloid, suppresses the activation of NF-κB and increases the expression of Klotho [67].
The 3-hydroxy-3 methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors such as statins (e.g. atorvastatin, rosuvastatin, and pitavastatin) also have anti-inflammatory and antioxidant effects. Statins promote Kl gene expression by activation of the FOXO signaling pathway and inhibition of the Rho/Rho-kinase pathway [32,33], or by modulating Nrf2 and Nrf2/HO-1 [68]. Pravastatin, in a dose-dependent manner, markedly increases Klotho expression, which is responsible for the benefits of statin therapy on endothelial dysfunction and atherosclerosis [5].
Finally, other mechanisms have been suggested. For instance, a regulatory role of peroxisome proliferator-activated receptor gamma-activated receptor-γ (PPAR-γ) on Kl has been proposed [69]. Thus, PPAR-γ agonists or thiazolidinediones such as ciglitazone, troglitazone, pioglitazone and rosiglitazone, could elevate Kl levels [49]. Another proposal has been the use of the antibiotic rapamycin, which is an mTOR inhibitor that increases Klotho expression. Certainly, a delay in the onset of age-related diseases has been described after rapamycin treatment [70].
Drugs capable of controlling epigenetic modifications such as those modifying Histone Acetyltransferase (HAT) and Histone Deacetylase (HDAC) enzymes should not be overlooked in this list. Indeed, the HDAC inhibitor trichostatin-A induces a positive regulation of Kl [71] while methylation of the Kl gene promoter silences its gene expression [71]. Therefore, DNA methyltransferase inhibitors, such as azacitidine, decitabine, and zebularine, could have therapeutic potential as a Kl upregulation approach.
Klotho-induced neuroprotection
The CNS is the second most abundant organ expressing Kl after the kidneys [72]. Both Kl mRNA and protein are present in the brain parenchyma, colocalizing in neurons and oligodendrocytes. The highest amount of brain Kl was detected in the choroid plexus and is expressed by ependymal cells. Kl can be found as well in the cortex, cerebellum, hippocampus, striatum, substantia nigra, medulla, olfactory bulb, and different limbic areas, such as the thalamus, hypothalamus, and nuclei of the amygdala [73]. As for intracellular distribution, Kl has been detected in the soma and dendrites of hippocampal neurons. The roles of Kl in the nervous system are not fully understood but Kl may play an important role in neuroprotection [74,75]. Overall, Kl is necessary for healthy and normal brain function throughout life.
So far, the main conclusion obtained from the first published work in mice homozygous for a hypomorphic Klotho gene (kl/KL) was that the Klotho protein has anti-aging properties. Klotho may be part of the neuronal degeneration process [5] and is involved in the regulation of brain aging. This is suggested by the impaired cognition and abnormal brain pathology observed in Klotho mutant mice [76] and by the analysis of the genetic profile of aging changes in the white matter of the rhesus monkey brain [77]. In fact, a correlation between low Klotho levels and increased risk of stroke has been described. Klotho may also activate antiaging signaling pathways such as antioxidative and anti-inflammatory pathways.
A recent study showed a positive correlation between CSF Klotho levels with the stage of the disease. However, only one study has determined CSF Klotho levels in AD patients. This study showed a similar decrease in Klotho compared with controls [47]. How Klotho is related to AD progression in the human brain remains unknown, but studies in mice suggest that Klotho modulates N-methyl-d-aspartate receptor function and activates microglial cells to promote cognitive function [47].
Klotho depletion is associated with nerve damage and brain dysfunction [12,75], with synaptic destruction, axonal transport impairment, nerve fiber impairment, and nerve degeneration [4,76]. Mice lacking the KL gene have been shown to have learning and remembering problems, possibly due to reduced hippocampal synapses, axonal transport disorders, and hippocampal nerve damage [76]. Kl has also been shown to improve long-term potentiation (LTP) by inducing synaptic NMDA receptors and related genes such as FOS in the hippocampus and cortex, leading to learning and memory improvement [78].
Kl can bind to soluble amyloid precursor protein (APPsβ) and thus, might prevent the formation of β-amyloid structures, protecting the CNS against amyloid toxicity [79] . Semba et al. showed that CSF Kl levels were lower in AD patients than in healthy individuals, and higher in younger than in older people [80]. Existing studies with amyloidogenic mouse models have shown that overexpression of Klotho protein in the brain can ameliorate AD-like pathology and cognitive impairment as well as reverse neuronal damage. It is also known that Klotho can ameliorate Aβ accumulation in these murine models by regulating Aβ-related transporters and microglia transformation. In the early stages of AD, degradation pathways, i.e., autophagy, the Ubiquitin-Proteasome System (UPS), and Chaperone-Mediated Autophagy (CMA) are impaired. More importantly, the accumulation of β amyloid causes dysfunction in the lysosome and the Lysosomal Autophagy Pathway (LAP), leading to neuronal loss [81]. Recent studies have shown that Klotho expression and autophagy are related to the anatomical pathology of AD. Overexpression of Klotho can promote LAP in AD through activation of the beclin1 pathway [82].
Klotho displays anti-inflammatory actions under pathological conditions [83]. This is likely to take place by the blockade of the signaling pathway of NFκB. Human CNS contains a highly active NF-κB signaling system with deep implications for neurological health [84,85]. NF-κB regulates a family of microRNAs (miRNAs) which includes miRNA-9, miRNA-30b, miRNA-34a, miRNA-146a, and miRNA-155. These miRNAs, in addition to having a general role in immunity, inflammation, and gene function in the CNS [86-88], are involved in the neurodegenerative pathogenesis of AD. Indeed, these miRNAs have been found to be significantly upregulated in this condition [89,90]. In line with these findings, overexpression of α-Klotho in the mouse choroid plexus ameliorates behavioral shortage and increases the number of living neurons upon brain hypoperfusion. All this is accompanied by a decrease in the production of proinflammatory cytokines and activation of astrocytes and microglia [43]. Oxidative stress generates mitochondrial dysfunction, impairs DNA repair, and causes cell damage. All the latter are part of the pathogenesis of neurodegenerative diseases, such as AD [91]. Thus, Klotho activates Nrf2 in a way that protects from renal, cardiovascular, and neurological disease [92,93].
Other pathways, such as the Insulin/IGF pathway, have been related to Klotho in the CNS. Insulin/IGF signaling promotes olfactory associative learning [94,95] and is involved in CNS plasticity in the hypothalamus, hippocampus, olfactory bulb, and other brain areas [27,96]. Alterations in the insulin signaling cascade underlie cognitive impairment and the development of several neurodegenerative diseases. Blockade of IGF-I receptors has been associated with cerebral amyloidosis and accumulation of hyperphosphorylated tau protein. Also, cognitive impairment and other neuropathological changes are typical of AD [97]. In addition, the blockade of IGF-I signaling induced by s-Klotho has been shown to increase resistance to oxidative stress, thereby improving survival rates [28].
Another target pathway is Wnt. Wnt is expressed in various CNS territories such as radial glia, oligodendrocytes, microglia, astrocytes, and neurons [98-100] where it regulates neuronal patterning, stem cell proliferation, and neurogenesis [101-103]. Wnt, by binding to the so-called Frizzled receptors, engages in systemic physiological processes such as cell differentiation, polarity, and migration [101,104,105]. Wnt also protects against β-amyloid peptide (Aβ) neurotoxicity. This seems to be due to the role Wnt takes in glycogen synthase kinase-3 β (GSK-3 β)-catalyzed Tau phosphorylation. A hyperactivation of Wnt is associated with the generation of embryonic degenerative abnormalities and tissue fibrosis [29,105-107]. Wnt dysfunction may be involved in aging and has been associated with memory impairment [98,99,101,103,105-107] and in learning and memory processes [106-108]. In Klotho-deficient mice, excess Wnt activation promotes cellular senescence and has a negative impact on stem cell survival [109].
With regard to the relationship between FGF23 and Klotho, elevated levels of FGF23 are associated with Klotho deficiency [4]. Thus, the production of FGF23 is inversely related to the serum concentration of Klotho. This has been confirmed in the CNS as well. Indeed, a higher serum FGF23 concentration is associated with an increased risk of incident dementia and AD [110,111], perhaps due to the existence of cardiovascular risk factors or vascular evidence of brain injury or to an FGF23-dependent reduction in vitamin D levels, which is known to be a predictor of cognitive impairment in adults.
Finally, it should be emphasized that there is a serious lack of human studies focused on Klotho’s expression and functionality in the central nervous system. This is striking and therefore it is urgent to address this research, the results of which may be more than promising.
In conclusion, our understanding of Klotho and its function related to neurodegenerative disease progression is far from complete. However, it appears clear that Klotho offers protective roles against nervous system damage by interfering with numerous pathways. At present, one of the main limitations we still suffer from is the lack of specific klotho-modulating drugs. So, the effects on klotho-mediated protection of existing drugs should be further investigated.
- Avan A, Hachinski V. Global, regional, and national trends of dementia incidence and risk factors, 1990-2019: A Global Burden of Disease study. Alzheimers Dement. 2023 Apr;19(4):1281-1291. doi: 10.1002/alz.12764. Epub 2022 Aug 31. PMID: 36044376.
- Javidnia S, Cranwell S, Mueller SH, Selman C, Tullet JMA, Kuchenbaecker K, Alic N. Mendelian randomization analyses implicate biogenesis of translation machinery in human aging. Genome Res. 2022 Feb;32(2):258-265. doi: 10.1101/gr.275636.121. Epub 2022 Jan 25. PMID: 35078808; PMCID: PMC8805714.
- Franceschi C, Garagnani P, Morsiani C, Conte M, Santoro A, Grignolio A, Monti D, Capri M, Salvioli S. The Continuum of Aging and Age-Related Diseases: Common Mechanisms but Different Rates. Front Med (Lausanne). 2018 Mar 12;5:61. doi: 10.3389/fmed.2018.00061. PMID: 29662881; PMCID: PMC5890129.
- Kuro-o M, Matsumura Y, Aizawa H, Kawaguchi H, Suga T, Utsugi T, Ohyama Y, Kurabayashi M, Kaname T, Kume E, Iwasaki H, Iida A, Shiraki-Iida T, Nishikawa S, Nagai R, Nabeshima YI. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature. 1997 Nov 6;390(6655):45-51. doi: 10.1038/36285. PMID: 9363890.
- Pan HC, Chou KM, Lee CC, Yang NI, Sun CY. Circulating Klotho levels can predict long-term macrovascular outcomes in type 2 diabetic patients. Atherosclerosis. 2018 Sep;276:83-90. doi: 10.1016/j.atherosclerosis.2018.07.006. Epub 2018 Jul 17. PMID: 30048945.
- Matsumura Y, Aizawa H, Shiraki-Iida T, Nagai R, Kuro-o M, Nabeshima Y. Identification of the human klotho gene and its two transcripts encoding membrane and secreted klotho protein. Biochem Biophys Res Commun. 1998 Jan 26;242(3):626-30. doi: 10.1006/bbrc.1997.8019. PMID: 9464267.
- Janson L, Pettersson U. Cooperative interactions between transcription factors Sp1 and OTF-1. Proc Natl Acad Sci U S A. 1990 Jun;87(12):4732-6. doi: 10.1073/pnas.87.12.4732. PMID: 2191301; PMCID: PMC54191.
- Deary IJ, Harris SE, Fox HC, Hayward C, Wright AF, Starr JM, Whalley LJ. KLOTHO genotype and cognitive ability in childhood and old age in the same individuals. Neurosci Lett. 2005 Apr 11;378(1):22-7. doi: 10.1016/j.neulet.2004.12.005. Epub 2005 Jan 22. PMID: 15763166.
- Lindberg K, Amin R, Moe OW, Hu MC, Erben RG, Östman Wernerson A, Lanske B, Olauson H, Larsson TE. The kidney is the principal organ mediating klotho effects. J Am Soc Nephrol. 2014 Oct;25(10):2169-75. doi: 10.1681/ASN.2013111209. Epub 2014 May 22. PMID: 24854271; PMCID: PMC4178446.
- Andrukhova O, Zeitz U, Goetz R, Mohammadi M, Lanske B, Erben RG. FGF23 acts directly on renal proximal tubules to induce phosphaturia through activation of the ERK1/2-SGK1 signaling pathway. Bone. 2012 Sep;51(3):621-8. doi: 10.1016/j.bone.2012.05.015. Epub 2012 May 27. PMID: 22647968; PMCID: PMC3419258.
- Martín-Núñez E, Pérez-Castro A, Tagua VG, Hernández-Carballo C, Ferri C, Pérez-Delgado N, Rodríguez-Ramos S, Cerro-López P, López-Castillo Á, Delgado-Molinos A, López-Tarruella VC, Arévalo-Gómez MA, González-Luis A, Martín-Olivera A, Morales-Estévez CC, Mora-Fernández C, Donate-Correa J, Navarro-González JF. Klotho expression in peripheral blood circulating cells is associated with vascular and systemic inflammation in atherosclerotic vascular disease. Sci Rep. 2022 May 19;12(1):8422. doi: 10.1038/s41598-022-12548-z. PMID: 35590090; PMCID: PMC9120199.
- Cararo-Lopes MM, Mazucanti CHY, Scavone C, Kawamoto EM, Berwick DC. The relevance of α-KLOTHO to the central nervous system: Some key questions. Ageing Res Rev. 2017 Jul;36:137-148. doi: 10.1016/j.arr.2017.03.003. Epub 2017 Mar 18. PMID: 28323064.
- Kuro-O M. The Klotho proteins in health and disease. Nat Rev Nephrol. 2019 Jan;15(1):27-44. doi: 10.1038/s41581-018-0078-3. PMID: 30455427.
- Martin A, David V, Quarles LD. Regulation and function of the FGF23/klotho endocrine pathways. Physiol Rev. 2012 Jan;92(1):131-55. doi: 10.1152/physrev.00002.2011. PMID: 22298654; PMCID: PMC3306265.
- Quarles LD. Role of FGF23 in vitamin D and phosphate metabolism: implications in chronic kidney disease. Exp Cell Res. 2012 May 15;318(9):1040-8. doi: 10.1016/j.yexcr.2012.02.027. Epub 2012 Mar 7. PMID: 22421513; PMCID: PMC3336874.
- Kurosu H, Ogawa Y, Miyoshi M, Yamamoto M, Nandi A, Rosenblatt KP, Baum MG, Schiavi S, Hu MC, Moe OW, Kuro-o M. Regulation of fibroblast growth factor-23 signaling by klotho. J Biol Chem. 2006 Mar 10;281(10):6120-3. doi: 10.1074/jbc.C500457200. Epub 2006 Jan 25. PMID: 16436388; PMCID: PMC2637204.
- Shimada T, Mizutani S, Muto T, Yoneya T, Hino R, Takeda S, Takeuchi Y, Fujita T, Fukumoto S, Yamashita T. Cloning and characterization of FGF23 as a causative factor of tumor-induced osteomalacia. Proc Natl Acad Sci U S A. 2001 May 22;98(11):6500-5. doi: 10.1073/pnas.101545198. Epub 2001 May 8. PMID: 11344269; PMCID: PMC33497.
- Goetz R, Ohnishi M, Kir S, Kurosu H, Wang L, Pastor J, Ma J, Gai W, Kuro-o M, Razzaque MS, Mohammadi M. Conversion of a paracrine fibroblast growth factor into an endocrine fibroblast growth factor. J Biol Chem. 2012 Aug 17;287(34):29134-46. doi: 10.1074/jbc.M112.342980. Epub 2012 Jun 25. PMID: 22733815; PMCID: PMC3436551.
- Kužmová Z, Kužma M, Gažová A, Kovářová M, Jackuliak P, Killinger Z, Kyselovič J, Payer J. Fibroblast Growth Factor 23 and Klotho Are Associated With Trabecular Bone Score but Not Bone Mineral Density in the Early Stages of Chronic Kidney Disease: Results of the Cross-Sectional Study. Physiol Res. 2021 Nov 30;70(Suppl 1):S43-S51. doi: 10.33549/physiolres.934773. PMID: 34918528; PMCID: PMC8884384.
- Mikuła-Pietrasik J, Rutecki S, Książek K. The functional multipotency of transforming growth factor β signaling at the intersection of senescence and cancer. Cell Mol Life Sci. 2022 Mar 19;79(4):196. doi: 10.1007/s00018-022-04236-y. PMID: 35305149.
- Tominaga K, Suzuki HI. TGF-β Signaling in Cellular Senescence and Aging-Related Pathology. Int J Mol Sci. 2019 Oct 10;20(20):5002. doi: 10.3390/ijms20205002. PMID: 31658594; PMCID: PMC6834140.
- Prud'homme GJ, Kurt M, Wang Q. Pathobiology of the Klotho Antiaging Protein and Therapeutic Considerations. Front Aging. 2022 Jul 12;3:931331. doi: 10.3389/fragi.2022.931331. PMID: 35903083; PMCID: PMC9314780.
- Yuan Q, Ren Q, Li L, Tan H, Lu M, Tian Y, Huang L, Zhao B, Fu H, Hou FF, Zhou L, Liu Y. A Klotho-derived peptide protects against kidney fibrosis by targeting TGF-β signaling. Nat Commun. 2022 Jan 21;13(1):438. doi: 10.1038/s41467-022-28096-z. Erratum in: Nat Commun. 2022 Nov 4;13(1):6640. PMID: 35064106; PMCID: PMC8782923.
- Navarro-González JF, Donate-Correa J, Muros de Fuentes M, Pérez-Hernández H, Martínez-Sanz R, Mora-Fernández C. Reduced Klotho is associated with the presence and severity of coronary artery disease. Heart. 2014 Jan;100(1):34-40. doi: 10.1136/heartjnl-2013-304746. Epub 2013 Oct 28. PMID: 24165855.
- Kenyon C. The plasticity of aging: insights from long-lived mutants. Cell. 2005 Feb 25;120(4):449-60. doi: 10.1016/j.cell.2005.02.002. PMID: 15734678.
- Higashi Y, Sukhanov S, Anwar A, Shai SY, Delafontaine P. Aging, atherosclerosis, and IGF-1. J Gerontol A Biol Sci Med Sci. 2012 Jun;67(6):626-39. doi: 10.1093/gerona/gls102. Epub 2012 Apr 5. PMID: 22491965; PMCID: PMC3348497.
- Dyer AH, Vahdatpour C, Sanfeliu A, Tropea D. The role of Insulin-Like Growth Factor 1 (IGF-1) in brain development, maturation and neuroplasticity. Neuroscience. 2016 Jun 14;325:89-99. doi: 10.1016/j.neuroscience.2016.03.056. Epub 2016 Mar 31. PMID: 27038749.
- Yamamoto M, Clark JD, Pastor JV, Gurnani P, Nandi A, Kurosu H, Miyoshi M, Ogawa Y, Castrillon DH, Rosenblatt KP, Kuro-o M. Regulation of oxidative stress by the anti-aging hormone klotho. J Biol Chem. 2005 Nov 11;280(45):38029-34. doi: 10.1074/jbc.M509039200. Epub 2005 Sep 26. PMID: 16186101; PMCID: PMC2515369.
- Liu H, Fergusson MM, Castilho RM, Liu J, Cao L, Chen J, Malide D, Rovira II, Schimel D, Kuo CJ, Gutkind JS, Hwang PM, Finkel T. Augmented Wnt signaling in a mammalian model of accelerated aging. Science. 2007 Aug 10;317(5839):803-6. doi: 10.1126/science.1143578. PMID: 17690294.
- Wang Y, Sun Z. Current understanding of klotho. Ageing Res Rev. 2009 Jan;8(1):43-51. doi: 10.1016/j.arr.2008.10.002. Epub 2008 Oct 31. PMID: 19022406; PMCID: PMC2637560.
- Lawrence T, Gilroy DW, Colville-Nash PR, Willoughby DA. Possible new role for NF-kappaB in the resolution of inflammation. Nat Med. 2001 Dec;7(12):1291-7. doi: 10.1038/nm1201-1291. PMID: 11726968.
- Dinkova-Kostova AT, Kostov RV, Kazantsev AG. The role of Nrf2 signaling in counteracting neurodegenerative diseases. FEBS J. 2018 Oct;285(19):3576-3590. doi: 10.1111/febs.14379. Epub 2018 Jan 29. PMID: 29323772; PMCID: PMC6221096.
- Qu Z, Sun J, Zhang W, Yu J, Zhuang C. Transcription factor NRF2 as a promising therapeutic target for Alzheimer's disease. Free Radic Biol Med. 2020 Nov 1;159:87-102. doi: 10.1016/j.freeradbiomed.2020.06.028. Epub 2020 Jul 28. PMID: 32730855.
- Panda H, Wen H, Suzuki M, Yamamoto M. Multifaceted Roles of the KEAP1-NRF2 System in Cancer and Inflammatory Disease Milieu. Antioxidants (Basel). 2022 Mar 11;11(3):538. doi: 10.3390/antiox11030538. PMID: 35326187; PMCID: PMC8944524.
- Haga M, Okada M. Systems approaches to investigate the role of NF-κB signaling in aging. Biochem J. 2022 Jan 28;479(2):161-183. doi: 10.1042/BCJ20210547. PMID: 35098992; PMCID: PMC8883486.
- Pahl HL. Activators and target genes of Rel/NF-kappaB transcription factors. Oncogene. 1999 Nov 22;18(49):6853-66. doi: 10.1038/sj.onc.1203239. PMID: 10602461.
- Li ZW, Chu W, Hu Y, Delhase M, Deerinck T, Ellisman M, Johnson R, Karin M. The IKKbeta subunit of IkappaB kinase (IKK) is essential for nuclear factor kappaB activation and prevention of apoptosis. J Exp Med. 1999 Jun 7;189(11):1839-45. doi: 10.1084/jem.189.11.1839. PMID: 10359587; PMCID: PMC2193082.
- Li L, Wang Y, Gao W, Yuan C, Zhang S, Zhou H, Huang M, Yao X. Klotho Reduction in Alveolar Macrophages Contributes to Cigarette Smoke Extract-induced Inflammation in Chronic Obstructive Pulmonary Disease. J Biol Chem. 2015 Nov 13;290(46):27890-900. doi: 10.1074/jbc.M115.655431. Epub 2015 Sep 18. PMID: 26385922; PMCID: PMC4646031.
- Maekawa Y, Ishikawa K, Yasuda O, Oguro R, Hanasaki H, Kida I, Takemura Y, Ohishi M, Katsuya T, Rakugi H. Klotho suppresses TNF-alpha-induced expression of adhesion molecules in the endothelium and attenuates NF-kappaB activation. Endocrine. 2009 Jun;35(3):341-6. doi: 10.1007/s12020-009-9181-3. Epub 2009 Apr 15. PMID: 19367378.
- Zhao Y, Banerjee S, Dey N, LeJeune WS, Sarkar PS, Brobey R, Rosenblatt KP, Tilton RG, Choudhary S. Klotho depletion contributes to increased inflammation in kidney of the db/db mouse model of diabetes via RelA (serine)536 phosphorylation. Diabetes. 2011 Jul;60(7):1907-16. doi: 10.2337/db10-1262. Epub 2011 May 18. PMID: 21593200; PMCID: PMC3121423.
- Moreno JA, Izquierdo MC, Sanchez-Niño MD, Suárez-Alvarez B, Lopez-Larrea C, Jakubowski A, Blanco J, Ramirez R, Selgas R, Ruiz-Ortega M, Egido J, Ortiz A, Sanz AB. The inflammatory cytokines TWEAK and TNFα reduce renal klotho expression through NFκB. J Am Soc Nephrol. 2011 Jul;22(7):1315-25. doi: 10.1681/ASN.2010101073. Epub 2011 Jun 30. PMID: 21719790; PMCID: PMC3137579.
- Lin W, Wu X, Wen J, Fei Y, Wu J, Li X, Zhang Q, Dong Y, Xu T, Fan Y, Wang N. Nicotinamide retains Klotho expression and ameliorates rhabdomyolysis-induced acute kidney injury. Nutrition. 2021 Nov-Dec;91-92:111376. doi: 10.1016/j.nut.2021.111376. Epub 2021 Jun 2. PMID: 34274652.
- Zhou HJ, Li H, Shi MQ, Mao XN, Liu DL, Chang YR, Gan YM, Kuang X, Du JR. Protective Effect of Klotho against Ischemic Brain Injury Is Associated with Inhibition of RIG-I/NF-κB Signaling. Front Pharmacol. 2018 Jan 18;8:950. doi: 10.3389/fphar.2017.00950. PMID: 29403373; PMCID: PMC5778393.
- Martín-Núñez E, Donate-Correa J, Ferri C, López-Castillo Á, Delgado-Molinos A, Hernández-Carballo C, Pérez-Delgado N, Rodríguez-Ramos S, Cerro-López P, Tagua VG, Mora-Fernández C, Navarro-González JF. Association between serum levels of Klotho and inflammatory cytokines in cardiovascular disease: a case-control study. Aging (Albany NY). 2020 Jan 27;12(2):1952-1964. doi: 10.18632/aging.102734. Epub 2020 Jan 27. PMID: 31986490; PMCID: PMC7053623.
- Nie F, Wu D, Du H, Yang X, Yang M, Pang X, Xu Y. Serum klotho protein levels and their correlations with the progression of type 2 diabetes mellitus. J Diabetes Complications. 2017 Mar;31(3):594-598. doi: 10.1016/j.jdiacomp.2016.11.008. Epub 2016 Nov 12. PMID: 27916483.
- Qiao Y, Liu F, Peng Y, Wang P, Ma B, Li L, Si C, Wang X, Zhang M, Song F. Association of serum Klotho levels with cancer and cancer mortality: Evidence from National Health and Nutrition Examination Survey. Cancer Med. 2023 Jan;12(2):1922-1934. doi: 10.1002/cam4.5027. Epub 2022 Jul 16. PMID: 35841322; PMCID: PMC9883546.
- Grøntvedt GR, Sando SB, Lauridsen C, Bråthen G, White LR, Salvesen Ø, Aarsland D, Hessen E, Fladby T, Waterloo K, Scheffler K. Association of Klotho Protein Levels and KL-VS Heterozygosity With Alzheimer Disease and Amyloid and Tau Burden. JAMA Netw Open. 2022 Nov 1;5(11):e2243232. doi: 10.1001/jamanetworkopen.2022.43232. PMID: 36413367; PMCID: PMC9682425.
- Sosa-Pineda B, Chowdhury K, Torres M, Oliver G, Gruss P. The Pax4 gene is essential for differentiation of insulin-producing beta cells in the mammalian pancreas. Nature. 1997 Mar 27;386(6623):399-402. doi: 10.1038/386399a0. PMID: 9121556.
- Zhang H, Li Y, Fan Y, Wu J, Zhao B, Guan Y, Chien S, Wang N. Klotho is a target gene of PPAR-gamma. Kidney Int. 2008 Sep;74(6):732-9. doi: 10.1038/ki.2008.244. Epub 2008 Jun 11. PMID: 18547997.
- Zhang R, Zheng F. PPAR-gamma and aging: one link through klotho? Kidney Int. 2008 Sep;74(6):702-4. doi: 10.1038/ki.2008.382. PMID: 18756295.
- Marsell R, Krajisnik T, Göransson H, Ohlsson C, Ljunggren O, Larsson TE, Jonsson KB. Gene expression analysis of kidneys from transgenic mice expressing fibroblast growth factor-23. Nephrol Dial Transplant. 2008 Mar;23(3):827-33. doi: 10.1093/ndt/gfm672. Epub 2007 Oct 2. PMID: 17911089.
- Zhou Q, Lin S, Tang R, Veeraragoo P, Peng W, Wu R. Role of Fosinopril and Valsartan on Klotho Gene Expression Induced by Angiotensin II in Rat Renal Tubular Epithelial Cells. Kidney Blood Press Res. 2010;33(3):186-92. doi: 10.1159/000316703. Epub 2010 Jun 23. PMID: 20571281.
- Xu Y, Sun Z. Molecular basis of Klotho: from gene to function in aging. Endocr Rev. 2015 Apr;36(2):174-93. doi: 10.1210/er.2013-1079. Epub 2015 Feb 19. PMID: 25695404; PMCID: PMC4399270.
- Hsu SC, Huang SM, Lin SH, Ka SM, Chen A, Shih MF, Hsu YJ. Testosterone increases renal anti-aging klotho gene expression via the androgen receptor-mediated pathway. Biochem J. 2014 Dec 1;464(2):221-9. doi: 10.1042/BJ20140739. PMID: 25163025.
- Sárvári M, Kalló I, Hrabovszky E, Solymosi N, Rodolosse A, Vastagh C, Auer H, Liposits Z. Hippocampal Gene Expression Is Highly Responsive to Estradiol Replacement in Middle-Aged Female Rats. Endocrinology. 2015 Jul;156(7):2632-45. doi: 10.1210/en.2015-1109. Epub 2015 Apr 29. PMID: 25924104.
- Mizuno I, Takahashi Y, Okimura Y, Kaji H, Chihara K. Upregulation of the klotho gene expression by thyroid hormone and during adipose differentiation in 3T3-L1 adipocytes. Life Sci. 2001 May 18;68(26):2917-23. doi: 10.1016/s0024-3205(01)01092-x. PMID: 11411791.
- Chen CD, Podvin S, Gillespie E, Leeman SE, Abraham CR. Insulin stimulates the cleavage and release of the extracellular domain of Klotho by ADAM10 and ADAM17. Proc Natl Acad Sci U S A. 2007 Dec 11;104(50):19796-801. doi: 10.1073/pnas.0709805104. Epub 2007 Dec 3. PMID: 18056631; PMCID: PMC2148378.
- Lau WL, Leaf EM, Hu MC, Takeno MM, Kuro-o M, Moe OW, Giachelli CM. Vitamin D receptor agonists increase klotho and osteopontin while decreasing aortic calcification in mice with chronic kidney disease fed a high phosphate diet. Kidney Int. 2012 Dec;82(12):1261-70. doi: 10.1038/ki.2012.322. Epub 2012 Aug 29. PMID: 22932118; PMCID: PMC3511664.
- Vojinovic J. Vitamin D receptor agonists' anti-inflammatory properties. Ann N Y Acad Sci. 2014 May;1317:47-56. doi: 10.1111/nyas.12429. Epub 2014 Apr 22. PMID: 24754474.
- Adorini L, Penna G, Fibbi B, Maggi M. Vitamin D receptor agonists target static, dynamic, and inflammatory components of benign prostatic hyperplasia. Ann N Y Acad Sci. 2010 Apr;1193:146-52. doi: 10.1111/j.1749-6632.2009.05299.x. PMID: 20398021.
- Mitani H, Ishizaka N, Aizawa T, Ohno M, Usui S, Suzuki T, Amaki T, Mori I, Nakamura Y, Sato M, Nangaku M, Hirata Y, Nagai R. In vivo klotho gene transfer ameliorates angiotensin II-induced renal damage. Hypertension. 2002 Apr;39(4):838-43. doi: 10.1161/01.hyp.0000013734.33441.ea. PMID: 11967236.
- Yoon HE, Ghee JY, Piao S, Song JH, Han DH, Kim S, Ohashi N, Kobori H, Kuro-o M, Yang CW. Angiotensin II blockade upregulates the expression of Klotho, the anti-ageing gene, in an experimental model of chronic cyclosporine nephropathy. Nephrol Dial Transplant. 2011 Mar;26(3):800-13. doi: 10.1093/ndt/gfq537. Epub 2010 Sep 2. PMID: 20813770; PMCID: PMC3108350.
- Mitobe M, Yoshida T, Sugiura H, Shirota S, Tsuchiya K, Nihei H. Oxidative stress decreases klotho expression in a mouse kidney cell line. Nephron Exp Nephrol. 2005;101(2):e67-74. doi: 10.1159/000086500. Epub 2005 Jun 21. PMID: 15976510.
- Piao SG, Kang SH, Lim SW, Chung BH, Doh KC, Heo SB, Jin L, Li C, Yang CW. Influence of N-acetylcysteine on Klotho expression and its signaling pathway in experimental model of chronic cyclosporine nephropathy in mice. Transplantation. 2013 Jul 27;96(2):146-53. doi: 10.1097/TP.0b013e318296c9a9. PMID: 23765110.
- Jaturakan O, Buranakarl C, Dissayabutra T, Chaiyabutr N, Kijtawornrat A, Rungsipipat A. Changes of Klotho protein and Klotho mRNA expression in a hydroxy-L-proline induced hyperoxaluric rat model. J Vet Med Sci. 2017 Nov 17;79(11):1861-1869. doi: 10.1292/jvms.17-0340. Epub 2017 Sep 23. PMID: 28943597; PMCID: PMC5709566.
- Rui Y, Li S, Luan F, Li D, Liu R, Zeng N. Several Alkaloids in Chinese Herbal Medicine Exert Protection in Acute Kidney Injury: Focus on Mechanism and Target Analysis. Oxid Med Cell Longev. 2022 May 13;2022:2427802. doi: 10.1155/2022/2427802. PMID: 35602100; PMCID: PMC9122709.
- Li H, Chen W, Chen Y, Zhou Q, Xiao P, Tang R, Xue J. Neferine Attenuates Acute Kidney Injury by Inhibiting NF-κB Signaling and Upregulating Klotho Expression. Front Pharmacol. 2019 Oct 15;10:1197. doi: 10.3389/fphar.2019.01197. PMID: 31680971; PMCID: PMC6804424.
- Mansouri A, Reiner Ž, Ruscica M, Tedeschi-Reiner E, Radbakhsh S, Bagheri Ekta M, Sahebkar A. Antioxidant Effects of Statins by Modulating Nrf2 and Nrf2/HO-1 Signaling in Different Diseases. J Clin Med. 2022 Feb 27;11(5):1313. doi: 10.3390/jcm11051313. PMID: 35268403; PMCID: PMC8911353.
- Yamashita T, Yoshioka M, Itoh N. Identification of a novel fibroblast growth factor, FGF-23, preferentially expressed in the ventrolateral thalamic nucleus of the brain. Biochem Biophys Res Commun. 2000 Oct 22;277(2):494-8. doi: 10.1006/bbrc.2000.3696. PMID: 11032749.
- Zhao Y, Zhao MM, Cai Y, Zheng MF, Sun WL, Zhang SY, Kong W, Gu J, Wang X, Xu MJ. Mammalian target of rapamycin signaling inhibition ameliorates vascular calcification via Klotho upregulation. Kidney Int. 2015 Oct;88(4):711-21. doi: 10.1038/ki.2015.160. Epub 2015 Jun 10. PMID: 26061549.
- Zhu Y, Cao X, Zhang X, Chen Q, Wen L, Wang P. DNA methylation-mediated Klotho silencing is an independent prognostic biomarker of head and neck squamous carcinoma. Cancer Manag Res. 2019 Feb 12;11:1383-1390. doi: 10.2147/CMAR.S188415. PMID: 30863149; PMCID: PMC6388988.
- Nakao VW, Mazucanti CHY, de Sá Lima L, de Mello PS, de Souza Port's NM, Kinoshita PF, Leite JA, Kawamoto EM, Scavone C. Neuroprotective action of α-Klotho against LPS-activated glia conditioned medium in primary neuronal culture. Sci Rep. 2022 Nov 7;12(1):18884. doi: 10.1038/s41598-022-21132-4. PMID: 36344527; PMCID: PMC9640694.
- Zeldich E, Chen CD, Colvin TA, Bove-Fenderson EA, Liang J, Tucker Zhou TB, Harris DA, Abraham CR. The neuroprotective effect of Klotho is mediated via regulation of members of the redox system. J Biol Chem. 2014 Aug 29;289(35):24700-15. doi: 10.1074/jbc.M114.567321. Epub 2014 Jul 18. PMID: 25037225; PMCID: PMC4148892.
- Torbus-Paluszczak M, Bartman W, Adamczyk-Sowa M. Klotho protein in neurodegenerative disorders. Neurol Sci. 2018 Oct;39(10):1677-1682. doi: 10.1007/s10072-018-3496-x. Epub 2018 Jul 30. PMID: 30062646; PMCID: PMC6154120.
- Abraham CR, Mullen PC, Tucker-Zhou T, Chen CD, Zeldich E. Klotho Is a Neuroprotective and Cognition-Enhancing Protein. Vitam Horm. 2016;101:215-38. doi: 10.1016/bs.vh.2016.02.004. Epub 2016 Mar 22. PMID: 27125744.
- Shiozaki M, Yoshimura K, Shibata M, Koike M, Matsuura N, Uchiyama Y, Gotow T. Morphological and biochemical signs of age-related neurodegenerative changes in klotho mutant mice. Neuroscience. 2008 Apr 9;152(4):924-41. doi: 10.1016/j.neuroscience.2008.01.032. Epub 2008 Feb 5. PMID: 18343589.
- Duce JA, Podvin S, Hollander W, Kipling D, Rosene DL, Abraham CR. Gene profile analysis implicates Klotho as an important contributor to aging changes in brain white matter of the rhesus monkey. Glia. 2008 Jan 1;56(1):106-17. doi: 10.1002/glia.20593. PMID: 17963266.
- Dubal DB, Zhu L, Sanchez PE, Worden K, Broestl L, Johnson E, Ho K, Yu GQ, Kim D, Betourne A, Kuro-O M, Masliah E, Abraham CR, Mucke L. Life extension factor klotho prevents mortality and enhances cognition in hAPP transgenic mice. J Neurosci. 2015 Feb 11;35(6):2358-71. doi: 10.1523/JNEUROSCI.5791-12.2015. PMID: 25673831; PMCID: PMC4323521.
- Erickson CM, Schultz SA, Oh JM, Darst BF, Ma Y, Norton D, Betthauser T, Gallagher CL, Carlsson CM, Bendlin BB, Asthana S, Hermann BP, Sager MA, Blennow K, Zetterberg H, Engelman CD, Christian BT, Johnson SC, Dubal DB, Okonkwo OC. KLOTHO heterozygosity attenuates APOE4-related amyloid burden in preclinical AD. Neurology. 2019 Apr 16;92(16):e1878-e1889. doi: 10.1212/WNL.0000000000007323. Epub 2019 Mar 13. PMID: 30867273; PMCID: PMC6550504.
- Semba RD, Moghekar AR, Hu J, Sun K, Turner R, Ferrucci L, O'Brien R. Klotho in the cerebrospinal fluid of adults with and without Alzheimer's disease. Neurosci Lett. 2014 Jan 13;558:37-40. doi: 10.1016/j.neulet.2013.10.058. Epub 2013 Nov 7. PMID: 24211693; PMCID: PMC4037850.
- Nixon RA. The aging lysosome: An essential catalyst for late-onset neurodegenerative diseases. Biochim Biophys Acta Proteins Proteom. 2020 Sep;1868(9):140443. doi: 10.1016/j.bbapap.2020.140443. Epub 2020 May 13. PMID: 32416272; PMCID: PMC7388076.
- Fernández ÁF, Sebti S, Wei Y, Zou Z, Shi M, McMillan KL, He C, Ting T, Liu Y, Chiang WC, Marciano DK, Schiattarella GG, Bhagat G, Moe OW, Hu MC, Levine B. Disruption of the beclin 1-BCL2 autophagy regulatory complex promotes longevity in mice. Nature. 2018 Jun;558(7708):136-140. doi: 10.1038/s41586-018-0162-7. Epub 2018 May 30. Erratum in: Nature. 2018 Sep;561(7723):E30. PMID: 29849149; PMCID: PMC5992097.
- Zhang Q, Yin S, Liu L, Liu Z, Cao W. Rhein reversal of DNA hypermethylation-associated Klotho suppression ameliorates renal fibrosis in mice. Sci Rep. 2016 Oct 5;6:34597. doi: 10.1038/srep34597. PMID: 27703201; PMCID: PMC5050540.
- Kaltschmidt B, Kaltschmidt C. NF-kappaB in the nervous system. Cold Spring Harb Perspect Biol. 2009 Sep;1(3):a001271. doi: 10.1101/cshperspect.a001271. Erratum in: Cold Spring Harb Perspect Biol. 2010 Jan;2(1):a001271. PMID: 20066105; PMCID: PMC2773634.
- Christian F, Smith EL, Carmody RJ. The Regulation of NF-κB Subunits by Phosphorylation. Cells. 2016 Mar 18;5(1):12. doi: 10.3390/cells5010012. PMID: 26999213; PMCID: PMC4810097.
- Brennan S, Keon M, Liu B, Su Z, Saksena NK. Panoramic Visualization of Circulating MicroRNAs Across Neurodegenerative Diseases in Humans. Mol Neurobiol. 2019 Nov;56(11):7380-7407. doi: 10.1007/s12035-019-1615-1. Epub 2019 Apr 29. PMID: 31037649; PMCID: PMC6815273.
- Juźwik CA, S Drake S, Zhang Y, Paradis-Isler N, Sylvester A, Amar-Zifkin A, Douglas C, Morquette B, Moore CS, Fournier AE. microRNA dysregulation in neurodegenerative diseases: A systematic review. Prog Neurobiol. 2019 Nov;182:101664. doi: 10.1016/j.pneurobio.2019.101664. Epub 2019 Jul 26. PMID: 31356849.
- Yoon S, Kim SE, Ko Y, Jeong GH, Lee KH, Lee J, Solmi M, Jacob L, Smith L, Stickley A, Carvalho AF, Dragioti E, Kronbichler A, Koyanagi A, Hong SH, Thompson T, Oh H, Salazar de Pablo G, Radua J, Shin JI, Fusar-Poli P. Differential expression of MicroRNAs in Alzheimer's disease: a systematic review and meta-analysis. Mol Psychiatry. 2022 May;27(5):2405-2413. doi: 10.1038/s41380-022-01476-z. Epub 2022 Mar 9. Erratum in: Mol Psychiatry. 2022 Apr 8;: PMID: 35264731.
- Jauhari A, Singh T, Mishra S, Shankar J, Yadav S. Coordinated Action of miR-146a and Parkin Gene Regulate Rotenone-induced Neurodegeneration. Toxicol Sci. 2020 Aug 1;176(2):433-445. doi: 10.1093/toxsci/kfaa066. PMID: 32392329.
- Alexandrov P, Zhai Y, Li W, Lukiw W. Lipopolysaccharide-stimulated, NF-kB-, miRNA-146a- and miRNA-155-mediated molecular-genetic communication between the human gastrointestinal tract microbiome and the brain. Folia Neuropathol. 2019;57(3):211-219. doi: 10.5114/fn.2019.88449. PMID: 31588707.
- Salim S. Oxidative Stress and the Central Nervous System. J Pharmacol Exp Ther. 2017 Jan;360(1):201-205. doi: 10.1124/jpet.116.237503. Epub 2016 Oct 17. PMID: 27754930; PMCID: PMC5193071.
- Xing L, Guo H, Meng S, Zhu B, Fang J, Huang J, Chen J, Wang Y, Wang L, Yao X, Wang H. Klotho ameliorates diabetic nephropathy by activating Nrf2 signaling pathway in podocytes. Biochem Biophys Res Commun. 2021 Jan 1;534:450-456. doi: 10.1016/j.bbrc.2020.11.061. Epub 2020 Nov 27. PMID: 33256980.
- Xiang T, Luo X, Ye L, Huang H, Wu Y. Klotho alleviates NLRP3 inflammasome-mediated neuroinflammation in a temporal lobe epilepsy rat model by activating the Nrf2 signaling pathway. Epilepsy Behav. 2022 Mar;128:108509. doi: 10.1016/j.yebeh.2021.108509. Epub 2022 Jan 29. PMID: 35104732.
- de Tredern E, Rabah Y, Pasquer L, Minatchy J, Plaçais PY, Preat T. Glial glucose fuels the neuronal pentose phosphate pathway for long-term memory. Cell Rep. 2021 Aug 24;36(8):109620. doi: 10.1016/j.celrep.2021.109620. PMID: 34433052; PMCID: PMC8411112.
- Naganos S, Horiuchi J, Saitoe M. Mutations in the Drosophila insulin receptor substrate, CHICO, impair olfactory associative learning. Neurosci Res. 2012 May;73(1):49-55. doi: 10.1016/j.neures.2012.02.001. Epub 2012 Feb 10. PMID: 22342328.
- Ferrario CR, Reagan LP. Insulin-mediated synaptic plasticity in the CNS: Anatomical, functional and temporal contexts. Neuropharmacology. 2018 Jul 1;136(Pt B):182-191. doi: 10.1016/j.neuropharm.2017.12.001. Epub 2017 Dec 5. PMID: 29217283; PMCID: PMC5988909.
- Carro E, Trejo JL, Spuch C, Bohl D, Heard JM, Torres-Aleman I. Blockade of the insulin-like growth factor I receptor in the choroid plexus originates Alzheimer's-like neuropathology in rodents: new cues into the human disease? Neurobiol Aging. 2006 Nov;27(11):1618-31. doi: 10.1016/j.neurobiolaging.2005.09.039. Epub 2005 Nov 7. PMID: 16274856.
- Briona LK, Poulain FE, Mosimann C, Dorsky RI. Wnt/ß-catenin signaling is required for radial glial neurogenesis following spinal cord injury. Dev Biol. 2015 Jul 1;403(1):15-21. doi: 10.1016/j.ydbio.2015.03.025. Epub 2015 Apr 14. PMID: 25888075; PMCID: PMC4469497.
- Marchetti B. Wnt/β-Catenin Signaling Pathway Governs a Full Program for Dopaminergic Neuron Survival, Neurorescue and Regeneration in the MPTP Mouse Model of Parkinson's Disease. Int J Mol Sci. 2018 Nov 24;19(12):3743. doi: 10.3390/ijms19123743. PMID: 30477246; PMCID: PMC6321180.
- Wexler EM, Paucer A, Kornblum HI, Palmer TD, Geschwind DH. Endogenous Wnt signaling maintains neural progenitor cell potency. Stem Cells. 2009 May;27(5):1130-41. doi: 10.1002/stem.36. Erratum in: Stem Cells. 2009 Oct;27(10):2636. Plamer, Theodore D [corrected to Palmer, Theodore D]. PMID: 19418460; PMCID: PMC2782960.
- Inestrosa NC, Tapia-Rojas C, Cerpa W, Cisternas P, Zolezzi JM. WNT Signaling Is a Key Player in Alzheimer's Disease. Handb Exp Pharmacol. 2021;269:357-382. doi: 10.1007/164_2021_532. PMID: 34486097.
- Inestrosa NC, Arenas E. Emerging roles of Wnts in the adult nervous system. Nat Rev Neurosci. 2010 Feb;11(2):77-86. doi: 10.1038/nrn2755. Epub 2009 Dec 16. PMID: 20010950.
- Budnik V, Salinas PC. Wnt signaling during synaptic development and plasticity. Curr Opin Neurobiol. 2011 Feb;21(1):151-9. doi: 10.1016/j.conb.2010.12.002. Epub 2011 Jan 14. PMID: 21239163; PMCID: PMC3499977.
- Nusse R, Clevers H. Wnt/β-Catenin Signaling, Disease, and Emerging Therapeutic Modalities. Cell. 2017 Jun 1;169(6):985-999. doi: 10.1016/j.cell.2017.05.016. PMID: 28575679.
- Tan CW, Gardiner BS, Hirokawa Y, Smith DW, Burgess AW. Analysis of Wnt signaling β-catenin spatial dynamics in HEK293T cells. BMC Syst Biol. 2014 Apr 8;8:44. doi: 10.1186/1752-0509-8-44. PMID: 24712863; PMCID: PMC4108056.
- Hayat R, Manzoor M, Hussain A. Wnt signaling pathway: A comprehensive review. Cell Biol Int. 2022 Jun;46(6):863-877. doi: 10.1002/cbin.11797. Epub 2022 Apr 6. PMID: 35297539.
- Rim EY, Clevers H, Nusse R. The Wnt Pathway: From Signaling Mechanisms to Synthetic Modulators. Annu Rev Biochem. 2022 Jun 21;91:571-598. doi: 10.1146/annurev-biochem-040320-103615. Epub 2022 Mar 18. PMID: 35303793.
- Tapia-Rojas C, Inestrosa NC. Wnt signaling loss accelerates the appearance of neuropathological hallmarks of Alzheimer's disease in J20-APP transgenic and wild-type mice. J Neurochem. 2018 Feb;144(4):443-465. doi: 10.1111/jnc.14278. Epub 2018 Jan 26. PMID: 29240990.
- Bian A, Neyra JA, Zhan M, Hu MC. Klotho, stem cells, and aging. Clin Interv Aging. 2015 Aug 4;10:1233-43. doi: 10.2147/CIA.S84978. PMID: 26346243; PMCID: PMC4531025.
- Wright CB, Shah NH, Mendez AJ, DeRosa JT, Yoshita M, Elkind MS, Sacco RL, DeCarli C, Rundek T, Silverberg S, Dong C, Wolf M. Fibroblast Growth Factor 23 Is Associated With Subclinical Cerebrovascular Damage: The Northern Manhattan Study. Stroke. 2016 Apr;47(4):923-8. doi: 10.1161/STROKEAHA.115.012379. Epub 2016 Mar 8. PMID: 26956260; PMCID: PMC4811730.
- Drew DA, Tighiouart H, Scott TM, Lou KV, Fan L, Shaffi K, Weiner DE, Sarnak MJ. FGF-23 and cognitive performance in hemodialysis patients. Hemodial Int. 2014 Jan;18(1):78-86. doi: 10.1111/hdi.12100. Epub 2013 Oct 24. PMID: 24164913; PMCID: PMC4443906.